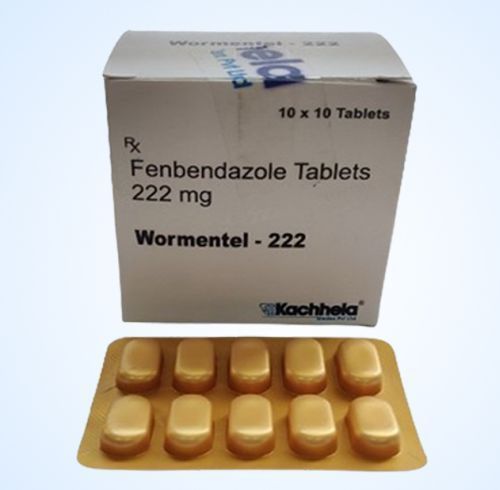
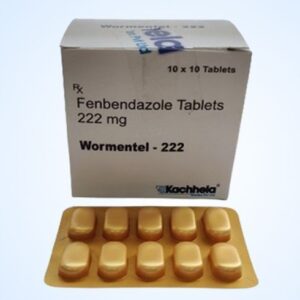
Fenbendazole 222 Mg
Brand Name : Fenbendazole 222 Mg
Active Ingredient (Generic Name) : Fenbendazole
Indication : Intestinal Parasites
Manufacturer : Kachhela Medex Pvt. Ltd.
Packaging : 10 tablets in 1 strip
Strength : 222 mg
What is Fenbendazole?
-
Fenbendazole is a benzimidazole anthelmintic (dewormer) used primarily in veterinary medicine to treat internal parasites in animals.
-
Its IUPAC name is methyl N-(6-phenylsulfanyl-1H-benzimidazol-2-yl)carbamate.
-
CAS number: 43210-67-9
-
In veterinary preparations (e.g. “Panacur”), one common formulation is granules of 22.2 % (which corresponds to 222 mg per gram)
Mechanism of Action
-
Fenbendazole works by binding to tubulin in parasite cells, thereby interfering with microtubule assembly. This disrupts the parasite’s cellular structure, nutrient absorption, and energy metabolism, leading to death of the parasite.
-
It tends to damage parasites more than host because of differences in tubulin affinity and lower absorption of the drug in host tissues.
-
After ingestion, fenbendazole is metabolized in the liver, producing metabolites such as oxfendazole which also have anthelmintic activity.
Veterinary Uses & Dosage
Fenbendazole is widely used in animals (dogs, cats, livestock) for treating internal parasites.
Common Indications in Animals
-
Roundworms (Ascarids)
-
Hookworms
-
Whipworms
-
Some species of tapeworms (e.g. certain Taenia)
-
In livestock, used for gastrointestinal nematodes, lungworms, and related parasites.
-
In dogs: used for mixed parasitic infections (hookworms, whipworms, roundworms, some tapeworms)
Dosing (Veterinary)
-
In Panacur® Granules 22.2% (a common product), the active content is 222 mg per gram of granule.
-
For dogs, one guideline is 50 mg/kg body weight per day, given for 3 consecutive days, for many internal parasites.
-
For carnivorous/omnivorous large animals (e.g. big cats, bears), Panacur uses 10 mg/kg daily for 3 days for some parasites.
-
In many dosing regimens, the drug is mixed with food to ensure ingestion.
Safety in Animals
-
Generally well tolerated in animals at labeled doses.
-
Mild gastrointestinal upset (vomiting, diarrhea, drooling) is among the more common side effects.
-
Rare side effects may include bone marrow suppression in some species (especially birds) at high doses.
-
In overdose, or in animals with high parasitic burden, there may be more serious reactions (allergic reactions, systemic stress) due to massive parasite death.
Use in Humans & “Off-Label / Alternative” Claims
Fenbendazole is not approved for human use by major regulatory agencies (FDA, EMA).
However, there has been anecdotal interest and early-stage research into using fenbendazole (and related benzimidazoles) for anticancer or antiparasitic effects in humans.
Research & Anecdotal Use
-
Some lab and animal studies suggest that fenbendazole may have anti-cancer activity (disrupting microtubules, altering energy metabolism in cancer cells).
-
A 2024 review titled “Oral Fenbendazole for Cancer Therapy in Humans and Animals” describes ongoing interest in repurposing it, but emphasizes that evidence in humans is preliminary.
-
On social media and alternative health communities, there are anecdotal reports of people self-administering 222 mg daily for cancer protocols. But such use is not medically endorsed or well studied.
Risks & Safety Concerns in Humans
Because human use is not well studied, risks are poorly defined, but there are documented case reports:
-
There have been case reports of drug-induced liver injury (hepatotoxicity) in patients taking fenbendazole for cancer off-label; liver injury resolved when drug was discontinued.
-
The poor oral bioavailability is a challenge — absorption from gut into circulation is limited, which may reduce its effect or necessitate high doses, raising toxicity risk.
-
Because fenbendazole can interfere with microtubules, theoretically there could be risks to rapidly dividing human cells (e.g. bone marrow, gastrointestinal lining) at higher doses, but data are lacking.
-
Interactions with other drugs are not well characterized in humans.
Reported Protocoled Dosing (Unverified / Anecdotal)
Some alternative-medicine sources propose:
-
222 mg daily for 3 days, followed by 4 days off (i.e. 3 on / 4 off), repeating cycles.
-
Some increases in dose (by increments of 222 mg) have been reported, up to some users targeting 1,000–2,000 mg/day — these are anecdotal and potentially dangerous.
-
Monitoring of liver enzymes and kidney function is often recommended in these protocols.
Key Parameters & Safety Summary for 222 mg Form
If one encounters a “fenbendazole 222 mg” product (which often corresponds to a veterinary formulation), here are important points:
-
This 222 mg is typically a measured dose in a capsule, tablet, or granule form (or in veterinary products, 222 mg per gram of powder)
-
Such a dose in humans is not standard or approved — any use must be seen as off-label and experimental.
-
The ingestion route is oral.
-
Adverse effects may include gastrointestinal upset, liver strain, allergies, or potentially more serious unrecognized risks at higher doses.
-
Because its use in humans is experimental, it is crucial to monitor liver and kidney function, blood counts, and other biomarkers if one chooses to try using it (under medical supervision).
-
Do not use in pregnancy, lactation, or in individuals with known liver disease unless under strict medical supervision (risk unknown).
| Tablet | 100 Tablet/s, 200 Tablet/s, 300 Tablet/s, 400 Tablet/s, 500 Tablet/s |
|---|




Reviews
There are no reviews yet.